What is sour service?
Sour service is referred to exploration and production environments in oil and gas that have enough H2S that could cause “CRACKING” of metallic materials.
The oil and gas industry recognize sour service as one of the toughest challenges in hydrocarbon production. H2S cracking is a risk to the integrity of assets and operations.
The exposure of certain pressurized metallic parts/components to this so-called ‘sour’ service has been known to initiate stress-induced cracking with often catastrophic failures of high-investment, critical processing equipment such as pressure safety relief valves (PSV/PRV) and other piping & pressure vessel parts.

In presence of water and H2S, “Hydrogen Atoms” are adsorbed and diffuse into the steel. They combine to form gaseous hydrogen which diffuses and could get trapped in the defect regions of the steel. Such accumulation of hydrogen will lead to cracks.
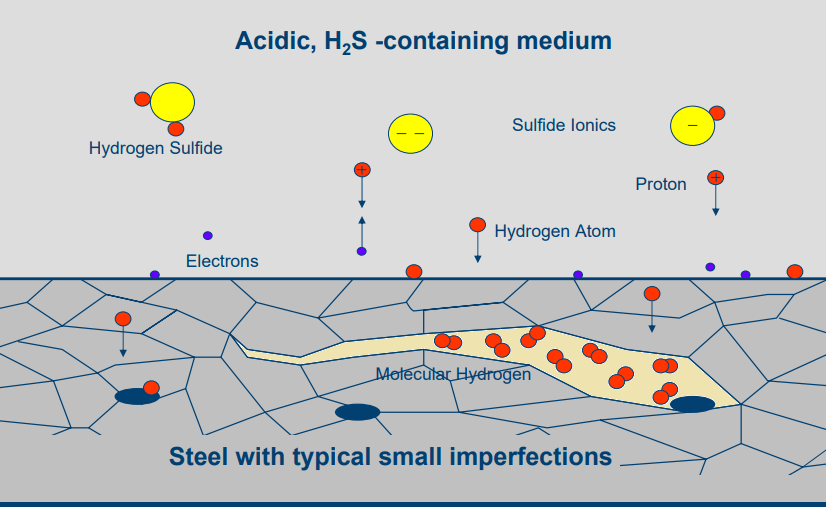
How many Cracking Types?
Here is all mechanisms of cracking that can be caused by H2S, including below cracking conditions:
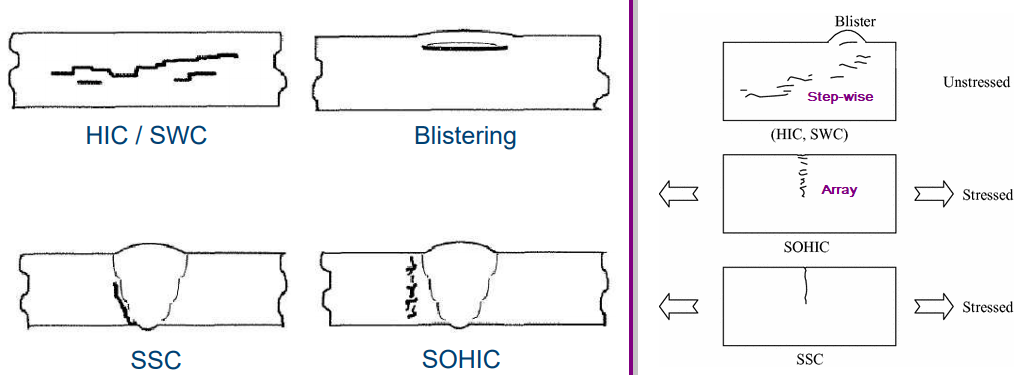
Sulfide stress cracking (SSC)
Stress corrosion cracking (SCC)
Step-wise cracking (SWC)
Soft zone cracking (SZC) ~ SOHIC appeared on Soft HAZ of weldment
Stress-oriented hydrogen-induced cracking (SOHIC)
Hydrogen-induced cracking (HIC)
Galvanically induced hydrogen stress cracking
Following the Cracks, the consequences of sudden failure of components used in the oil and gas industries have led to the creation of relevant engineering guidance documents.
Guess What?
All the industry bodies including API, IOGP as well as national codes ANSI, and international codes EN and ISO have recognized NACE MR0175 as a standard to prevent H2S cracking in metallic materials.

NACE MR0175 is an international standard that provides the requirements for metallic materials exposed to H2S in oil and gas production environments. NACE MR0175 is identical to ISO 15156. ANSI / NACE MR0175 / ISO 15156 lists the requirements for carbon steels, low alloys and corrosion resistant alloys.
Only in the USA is the MR0175 label still carried on the document as well as ISO15156 (the text is identical in all documents issued by individual Standards authorities). This is simply due to the need to retain documents supporting sour service requirements under certain USA State laws, which require facilities to comply with NACE MR0175. NACE fully supported the development and move to harmonies the EFC16 and EFC17 (European Federation of Corrosion publications N°16 and N°17) and NACE MR0175 documents and this has resulted in the production of ISO 15156, which is maintained via NACE and ISO. This replaces MR0175 in the just the same way that new versions of MR0175 replaced older versions.
The series ISO 15156 consisting of three (03) parts as below:
NACE MR0175/ISO 151546-1: General principles for selection of cracking resistant materials.
NACE MR0175/ISO 151546-2: Cracking-resistant carbon and low alloy steels.
NACE MR0175/ISO 151546-3: Cracking-resistant Corrosion Resistant Alloys (CRA’s) and other alloys.
What is the details requirement on Material selection, Testing, Chemical composition, Hardness on Steel Piping, Fitting, and Plate.
Please refer to next article.
What are the problem or concerns related to H2S ?
Toxic
First and far most important concern related to H2S is high toxicity. Human exposure of H2S at high concentration within a short period could lead to fatal.
Environment
H2S release to atmosphere would mix and dissolve in moisture and form acid rain which is corrosive and create human health concerns.
Corrosion & Stress Cracking
i) Sulphide Stress Corrosion Cracking (SSCC)
H2S dissolved in water to form weak acid promote corrosion and form free hydrogen. Free Hydrogen will penetrate the metal, reduce ductility of metal and potentially lead to stress failure below it yield stress, results Sulphide Stress Corrosion Cracking (SSCC).
Sulfide stress corrosion cracking (SSCC) is cracking of metal involving corrosion and tensile stress (residual and/or applied) in the presence of water and H2S. SSC is a form of hydrogen stress cracking (HSC) and involves embrittlement of the metal by atomic hydrogen that is produced by acid corrosion on the metal surface. Hydrogen uptake is promoted in the presence of sulfides. The atomic hydrogen can diffuse into the metal, reduce ductility and increase susceptibility to cracking. High strength metallic materials and hard weld zones are prone to SSC. (Source : NACE MR 0175)
Apart from SSCC, H2S also cause other cracking, including stress corrosion cracking (SCC), hydrogen-induced cracking (HIC) and stepwise cracking (SWC), stress-oriented hydrogen induced cracking (SOHIC), soft zone cracking (SZC) and galvanically induced hydrogen stress cracking.
Hydrogen Stress Cracking (HSC)
HSC refer to cracking that results from the presence of hydrogen in a metal and tensile stress (residual and/or applied). HSC describes cracking in metals that are not sensitive to SSC but which may be embrittled by hydrogen when galvanically coupled, as the cathode, to another metal that is corroding actively as an anode. The term galvanically induced HSC has been used for this mechanism of cracking.*
Stress Corrosion Cracking (SCC)
SCC refer to cracking of metal involving anodic processes of localized corrosion and tensile stress (residual and/or applied) in the presence of water and H2S. Chlorides and/or oxidants and elevated temperature can increase the susceptibility of metals to this mechanism of attacks. *
For Chlorides stress corrosion cracking (CSCC), read more in Chloride Stress Corrosion Cracking topic.
Hydrogen-Induced Cracking (HIC)
HIC refer to planar cracking that occurs in carbon and low alloy steels when atomic hydrogen diffuses into the steel and then combines to form molecular hydrogen at trap sites. Cracking results from the pressurization of trap sites by hydrogen. No externally applied stress is needed for the formation of hydrogen-induced cracks. Trap sites capable of causing HIC are commonly found in steels with high impurity levels that have a high density of planar inclusions and/or regions of anomalous microstructure (e.g. banding) produced by segregation of impurity and alloying elements in the steel. This form of hydrogen-induced cracking is not related to welding.*
Stepwise Cracking (SWC)
SWC refer to cracking that connects hydrogen-induced cracks on adjacent planes in a steel. This term describes the crack appearance. The linking of hydrogen-induced cracks to produce stepwise cracking is dependent upon local strain between the cracks and embrittlement of the surrounding steel by dissolved hydrogen. HIC/SWC is usually associated with low-strength plate steels used in the production of pipes and vessels.*
Stress-Oriented Hydrogen-Induced Cracking (SOHIC)
SOHIC refer to staggered small cracks formed approximately perpendicular to the principal stress (residual or applied) resulting in a “ladderlike” crack array linking (sometimes small) pre-existing HIC cracks. The mode of cracking can be categorized as SSC caused by a combination of external stress and the local strain around hydrogen-induced cracks. SOHIC is related to SSC and HIC/SWC. It has been observed in parent material of longitudinally welded pipe and in the heat-affected zone (HAZ) of welds in pressure vessels. SOHIC is a relatively uncommon phenomenon usually associated with low-strength ferritic pipe and pressure vessel steels.*
Soft Zone Cracking (SZC)
SZC refer to form of SSC that may occur when a steel contains a local “soft zone” of low yield strength material. Under service loads, soft zones may yield and accumulate plastic strain locally, increasing the SSC susceptibility to cracking of an otherwise SSC-resistant material. Such soft zones are typically associated with welds in carbon steels.*
* Source : NACE MR 0175 – ISO 15156


2 thoughts on “What is Sour Service in OIL & GAS?”