Stainless steel is known for excellent corrosion resistance. It is an integral part of modern life and is used in a range of applications, including heavy industry, architecture, automotive manufacture, surgery and dentistry. Stainless steel is an iron alloy with added elements such as chromium, nickel, silicon, manganese, nitrogen and carbon. The properties of the final alloy can be fine-tuned by altering the amounts of the various elements.
Now we will know How Stainless steel is made?
- Raw materials:
- Stainless steel is the most recycled material in the world and it is estimated that 82% of the stainless steel in use today will be recycled into new steel. When recycled, the stainless steel is not downgraded – recycled steel has the same quality and properties as the original steel. That’s why, on average, recycled steel makes up around 60% of the raw material used to make new stainless steel. To handle such large volumes of steel scrap, rigorous purchase routines are needed. At arrival, before entering the melt shop site, the steel scrap is tested to ensure that no radioactive components are present. It is then tested, analyzed, and sorted according to its alloying content to ensure that as little virgin material as possible is needed to get the right chemical composition of the stainless steel produced.
 2. The melt shop:
2. The melt shop:
- The raw materials are melted together in an electric arc furnace. It can take 8 to 12 hours of intense heat until the metal becomes molten. When the melting is finished, the molten steel is cast into semi-finished forms. These include blooms (rectangular shapes), billets (round or square shapes 1.5 inches or 3.8 centimeters in thickness), slabs, rods, and tube rounds.
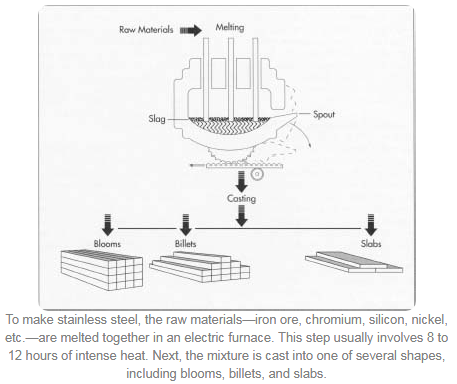
- The configuration of the melt shop depends on the capabilities of the steel works, but typically there are four main steps:
1) Melting of raw materials in an electric arc furnace (EAF)
2) Removal of carbon, sulfur, and possibly nitrogen, in a steel converter: The next stage is to remove excess carbon. This is done by processing the molten metal in an AOD (Argon Oxygen Decarburization) converter. The converter reduces the carbon by injecting an oxygen-argon mixture. At this stage, further alloying elements like nickel and molybdenum can be added to the AOD converter. Alternatively a VOD (Vacuum Oxygen Decarburization) converter can be used to when a very low carbon content is required.
3) Tuning of the steel composition and temperature in ladle treatment: Most stainless steels have exacting quality requirements. The tuning process allows fine adjustments to the chemical composition. Tuning is when the steel is slowly stirred to remove unwanted elements and improve consistency, while maintaining the required composition within the temperature limits.
4) Casting of slabs or ingots: The liquid steel ladle is then transported to the continuous casting machine or, in some cases, to the ingot casting area. Continuous casting is a process in which molten steel is converted into slabs of manageable size.
3. Hot rolling
– The hot rolling process begins at the reheat furnace where the slabs are heated to between 1100 and 1300°C, depending on the stainless steel grade. Hot rolling occurs at a temperature above the recrystallization temperature of the steel. The precise temperature depends on the desired stainless steel grade. The steel forms are heated and passed through high rolls. Blooms and billets are formed into bar and wire. Slabs are formed into plate, strip, and sheet.

4. Cold rolling, annealing, and pickling
- Cold rolling is used where extremely precise dimensions or an attractive finish are required. The process occurs below the recrystallization temperature of the steel. Cold rolling is carried out using small-diameter rolls and a series of supporting rolls. This process allows the production of wide sheets with improved surface finishes.
- Annealing is the process used to soften stainless steel, improve ductility, and refine grain structure. It is also used to relieve internal stresses in the metal caused by previous processing. During the annealing process the steel is heated and cooled under controlled conditions.
- Descaling/Pickling: The annealing process causes scale to form on the steel. These scales are commonly removed using pickling, which involves bathing the steel in nitric-hydrofluoric acid. Electrocleaning is an alternative method which uses an electric current to remove the scale.
5. Final processing
- Surface finish is important for stainless steel products, especially in applications where appearances are important. While most people are familiar with the look of stainless steel used for consumer products, there are actually a number of finishing options.
- Grinding wheels or abrasive belts are commonly used to grind or polish the steel. Other methods include buffing with cloth wheels with abrasive particles, dry etching using sandblasting, and wet etching using acid solutions. The smooth surface provides better corrosion resistance
- The final operations before sending the product to the customer are called finishing. This is where coils and plates are tailored to customer requirements, which can include:
- Leveling a plate or coil with a roller leveler or a stretch leveler, or a combination of both techniques
- Edge trimming of the coil to the desired width
- Slitting a coil into several narrow coils
- Cutting a coil into tailored or standard length sheets/plates in dedicated cut-to-length (CTL) lines
- Shearing or cutting into an order-sized square/rectangular plate or customized specially shaped plate from a larger quarto mother plate.
- Applying a plastic film coat to provide surface protection for further processing by the customer
- Edge preparation for special welding requirements
- Surface preparation such as grinding, brushing, pattern rolling, or embossing

- Process Diagram:

Summary: The importance of chromium in making stainless steel
Chromium is essential for the production of stainless steel; in fact there’s no viable alternative. Chromium is a hard, corrosion-resistant transition element that gives stainless steel its corrosion resistance. In general, the higher the chromium content, the more corrosion-resistant the steel.


One thought on “Stainless Steel Material: How is stainless steel made?”