Other Topic: Duplex piping and seawater
With this topic we will discuss about Corrosion resistance of Austenitic stainless steel in Sea water.
1) Sea water: Seawater is water from a sea or ocean.
Sea water in the world’s oceans has a salinity of approximately 3.5%, or 35 parts per thousand. This means that for every 1 litre (1000 mL) of seawater there are 35 grams of salts (mostly, but not entirely, sodium chloride) dissolved in it. Although a vast majority of seawater is found in oceans with salinity around 3.5%, seawater is not uniformly saline throughout the world.
2) Chlorides:
The recommended maximum level of chloride in U.S. drinking water is 250 mg/L.
Natural Chlorides constitute approximately 0.05% of the earth’s crust. Chloride concentrations of between 1 and 100 ppm (parts per million) are normal in freshwater. Seawater has a natural chloride concentration of 35,000 ppm.

3) Stainless steel:
Stainless Steel Material: what is stainless steel?
There are five families of stainless steel, each characterized by their structure:
- martensitic
- ferritic
- austenitic
- duplex (50/50 austenite/ferrite mixed structure)
- precipitation-hardened structures.
The families are characterized by their microstructures, which result from their particular compositions.
4) Why does Stainless steel prevent corrosion?
Stainless steel has chromium content is greater than about 12 percent by weight ( some document mention it is about 10.5% chromium ). Chromium provides corrosion resistance to these alloys by forming a thin, adherent, corrosion-resistant oxide film on a clean (e.g., pickled, wire/rotary brushed, or ground) surface (of the alloy).
When exposed to oxygen, whether in air or even in water, this layer will naturally form and will help to prevent corrosion of stainless steel beneath it. The effectiveness of this protective oxide layer can become compromised when the original oxide surface layer becomes damaged or scratched, but rapidly reforms its protective film in the presence of oxygen.
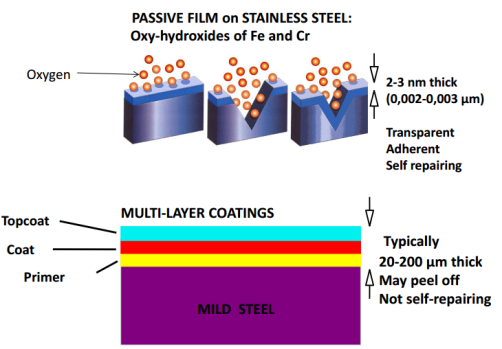
What happend when Protection layer is damaged? See below picture:

5) Why does Stainless steel corrosion?
The most forms of corrosion of stainless steel in seawater are crevice and pitting corrosion.
Both form of corrosion can be caused by the presence of Chloride ions in a solution and are also influenced by temperature and oxidation strength of the solution ( ex: chlorination… )
Why Stainless Steel Rust / Corrosion? – Part 1 – Pitting Corrosion!
Why Stainless steel rust / corrosion? – Part 2 – Other corrosion?
Types of corrosion on stainless steels:
a) Pitting
b) Crevice
c) Galvanic
d) Intergranular
e) Stress corrosion cracking
6) Conclusion:
We have some recommendation as below:
1. Stainless steel performance in seawater is good provided that proper precautions are being made and provided the proper alloy is being selected.
2. Because of (micro) biological activity and formation of a corrosive biofilm, natural seawater is significantly more corrosive than properly chlorinated seawater.
3. Cathodic protection improves performance of stainless steel in seawater and brackish water.
4. For 316 stainless steel, also severe atmospheric corrosion and corrosion under insulation may occur. So the advised materials selection for tubing is 6% Mo. It is not recommended to use stainless steel of the type AISI 316 or AISI 316L in stagnant seawater.
5) The traditional ferritic and austenitic stainless steels (including types 304 and 316) are unsuitable for use in seawater because they are prone to crevice corrosion and pitting attack, giving rise to a high probability of premature failure. Recent development work has produced materials with improved resistance to these forms of attack in seawater. The materials fall into three categories:
- a) Ferritic steels containing 25 to 29 % chromium, 3 to 4 % molybdenum with titanium or niobium for stabilisation and possibily up to 4 % nickel.
b) Austenitic steels containing about 18 to 25 % nickel, 20 % chromium, 6 % molybdenum and 0.1 to 0.2 % nitrogen.
c) Duplex structure steels containing 22 to 25 % chromium, 5 to 7% nickel, 3 % molybdenum and 0.15 to 0.2 % nitrogen.
6) Consider when selecting stainless steel for chlorinated waters with up to 500 ppm of chlorides:
• In systems with crevices present 304L and LDX 2101 are likely to suffer from crevice corrosion in water with a chloride content of 200 ppm and a temperature of 30°C, even at residual chlorine levels as low as 0.2 ppm.
• 316L may be useful in water with up to to 500 ppm at 30°C but with a significant risk of crevice corrosion if the residual chlorine concentration is 0.5 ppm or higher.
• 304L, LDX 2101® and 316L can all tolerate somewhat more corrosive conditions in systems which do not contain obvious crevices, provided all welds are of good quality and well cleaned.
• LDX 2101® is slightly more resistant than 304L in systems without severe crevices
• Residual chlorine levels as high as 1 ppm significantly increases the risk of pitting and crevice corrosion.
• LDX 2404® and 2205 show good resistance to both pitting and crevice corrosion in waters with ≤500 ppm of chlorides, ≤1 ppm chlorine and ≤50°C Consider when selecting stainless steel for chlorinated seawater:
• 2205 or lower alloyed grades are usually not sufficiently corrosion resistant.
• 6Mo superaustenitic, 25 Cr superduplex or higher alloyed grades are required
• In systems containing crevices 30°C is a maximum limit for 6Mo and superduplex grades and the level of chlorination needs to be carefully controlled, preferably the residual chlorine level should not exceed 0.5 ppm.
• 6Mo superaustenitic grades may tolerate somewhat more corrosive conditions, e.g. a temperature somewhat exceeding 30º C in “crevice free systems”, while 25Cr superduplex may suffer pitting in weldments.
• 654 SMO® is an option for systems with higher temperatures or residual chlorine levels.
Pls refer to other topic:
Stainless Steel Material: How is stainless steel made?
Stainless steel material: what difference between stainless steel and carbon steel?


One thought on “Stainless Steel Material In Sea Water”