Summary the important notes on this concept:
- SS will be 885*F (475*C) embrittlement and, or sigma-phase promotion by Ferrite content.
- The ferrite contained in Austenite SS is kept around 2-5% for weldability.
- The Embrittlement by Sigma phase is prevented by keep FN < 10
- The Embrittlement by 475*C is prevented by keeping FN <14
- Example: Some refinery specs required FN on SS347 <9 to prevent the Embrittlement (475*C Embrittle + Sigma phase embrittle)
There are a number of different types of steels that may be referred to as ‘stainless’; previous articles have considered ferritic and precipitation hardening steels for example. It is therefore advisable to be specific and to refer to the group to which the steel belongs in order to avoid confusion. Although commonly referred to as ‘stainless steel’, the steels covered in this article should be more correctly referred to as austenitic, 18/8 or chromium-nickel stainless steels.
As with the other types of stainless steels, the austenitic stainless steels are corrosion and oxidation resistant due to the presence of chromium that forms a self-healing protective film on the surface of the steel. They also have very good toughness at extremely low temperatures so are used extensively in cryogenic applications. They can be hardened and their strength increased by cold-working but not by heat treatment. They are the most easily weldable of the stainless steel family and can be welded by all welding processes, the main problems being the avoidance of hot cracking and the preservation of corrosion resistance.
A convenient and commonly used shorthand identifying the individual alloy within the austenitic stainless steel group is the ASTM system. This uses a three-digit number ‘3XX’, the ‘3’ identifying the steel as an austenitic stainless, and with additional letters to identify the composition and certain characteristics of the alloy eg type 304H, type 316L etc; this ASTM method will be used in this article.
Typical compositions of some of the alloys are given in Table 1. The type 304 grade may be regarded as the archetypal austenitic stainless steel from which the other grades are derived and changes in composition away from that of type 304 result in a change in the identification number and are highlighted in red.
Table 1 Typical compositions of some austenitic stainless steel alloys
| ASTM No. (type) |
Composition wt% | Microstructure | ||||||
|---|---|---|---|---|---|---|---|---|
| C (max) | Si (max) | Mn (max) | Cr | Ni | Mo | Others | Austenite – A Ferrite – F |
|
| 304 | 0.08 | 0.75 | 2.0 | 18/20 | 8/11 | – | – | A+2/8%F |
| 304L | 0.035 | 0.75 | 2.0 | 18/20 | 8/11 | – | – | A + 2/8%F |
| 304H | 0.04 – 0.10 | 0.75 | 2.0 | 18/20 | 8/11 | – | – | A + 2/8%F |
| 304N | 0.08 | 0.75 | 2.0 | 18/20 | 8/11 | – | 0.1/0.16N | A + 2/8%F |
| 316 | 0.08 | 0.75 | 2.0 | 16/18 | 11/14 | 2/3 | – | A + 3/10%F |
| 347 | 0.08 | 0.75 | 2.0 | 17/20 | 9/13 | – | Nb : 10xC | A + 4/12%F |
| 321 | 0.08 | 0.75 | 2.0 | 17/19 | 9/12 | – | Ti: 5xC | A + 4/12%F |
| 310 | 0.15 | 0.75 | 2.0 | 24/26 | 19/22 | – | – | 100% A |
| 309 | 0.08 | 1.0 | 2.0 | 22/24 | 12/15 | – | – | A + 8/15%F |
| 308L (generally filler metal only) | 0.03 | 1.0 | 2.0 | 19/21 | 10/12 | A + 4/12%F | ||
The 3XX may be followed by a letter that gives more information about the specific alloy as shown in the Table. ‘L’ is for a low carbon austenitic stainless steel for use in an aggressive corrosive environment; ‘H’ for high carbon steel with improved high-temperature strength for use in creep applications; ‘N’ for nitrogen-bearing steel where a higher tensile strength than conventional steel is required. These suffixes are used with most of the alloy designations eg type 316L, type 316LN, type 347H, where the composition has been modified from that of the base alloy.
Austenitic stainless steels are metallurgically simple alloys. They are either 100% austenite or austenite with a small amount of ferrite (see Table 1). This is not the ferrite to be found in carbon steel but a high-temperature form known as delta (δ) -ferrite. Unlike carbon and low alloy steels the austenitic stainless steels undergo no phase changes as they cool from high temperatures. They cannot therefore be quench hardened to form martensite and their mechanical properties to a great extent are unaffected by welding. Cold (hydrogen-induced) cracking is therefore not a problem and preheat is not necessary irrespective of component thickness.
Alloying elements in an austenitic stainless steel can be divided into two groups; those that promote the formation of austenite and those that favour the formation of ferrite. The main austenite formers are nickel, carbon, manganese and nitrogen; the important ferrite formers are chromium, silicon, molybdenum and niobium. By varying the amounts of these elements, the steel can be made to be fully austenitic or can be designed to contain a small amount of ferrite; the importance of this will be discussed later.
In 1949 Anton Schaeffler published a constitutional or phase diagram that illustrates the effects of composition on the microstructure. In the diagram Schaeffler assigned a factor to the various elements, the factor reflecting the strength of the effect on the formation of ferrite or austenite; these factors can be seen in the diagram. The elements are then combined into two groups to give chromium and nickel ‘equivalents’. These form the x and y axes of the diagram and, knowing the composition of austenitic stainless steel, enables the proportions of the phases to be determined.
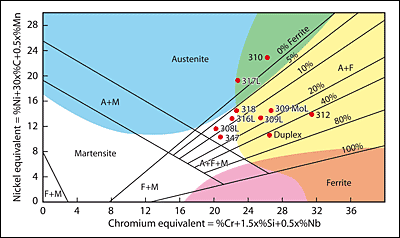
Typical positions of some of the commoner alloys are given in Fig.1. Also superimposed on this diagram are coloured areas identifying some of the fabrication problems that may be encountered with austenitic stainless steels.
Although all the austenitic stainless steels are sensitive to hot cracking, the fully austenitic steels falling within the vertically blue area in Fig.1 such as type 310 are particularly sensitive.
The main culprits are sulphur and phosphorus. To this end, these tramp elements have been progressively reduced such that steels with less than 0.010% sulphur and phosphorus less than 0.020% are now readily available. Ideally, a type 310 or type 317 alloy should have sulphur and phosphorus levels below some 0.003%. Cleanliness is also the most important and thorough degreasing must be carried out immediately prior to welding.
The steels such as type 304, type 316, type 347 that fall within, or close to, the small uncoloured triangular region in the centre of the diagram contain a small amount of delta-ferrite and, whilst not being immune to hot cracking, have improved resistance to the formation of sulphur-containing liquid films. The reasons for this are that a) ferrite can dissolve more sulphur and phosphorus than austenite so they are retained in solution rather than being available to form liquid films along the grain boundaries and b) the presence of quite a small amount of ferrite increases the grain boundary area such that any liquid films must spread over a greater area and can no longer form a continuous liquid film. The 100% austenitic steels do not have this advantage.
One problem that has arisen with very low sulphur steels is a phenomenon known as ‘cast to cast variation’ or ‘variable penetration’. The weld pool in a low sulphur steel (<0.005%) tends to be wide with shallow penetration; a steel with sulphur over some 0.010% has a narrower, more deeply penetrating weld bead.
This is generally only a problem with the use of the fully automated TIG welding process, a manual welder being capable of coping with the variations in penetration due to the differences in sulphur content in different casts of steel. However, automated TIG welding procedures developed on a ‘high’ sulphur steel, when used to weld a low sulphur steel may result in lack of penetration type defects; the reverse situation may result in excessive penetration.
Changes to the procedure that have mitigated, but never eliminated, this problem have included slow travel speed, pulsed current, use of Ar/H2 shield gas mixtures. Other methods include specifying minimum sulphur of, say, 0.010% or segregating the steels into batches with known penetration characteristics and developing welding procedures to suit. The A-TIG activated flux process has also been found to be of benefit.
The problems of welding the fully ferritic steels that fall into the pink area, where grain growth and embrittlement is a problem.
The austenitic stainless steels falling into the yellow area will also embrittle but this is as a result of the formation of hard brittle phases called ‘sigma’ (σ) and ‘chi’ (χ). This embrittlement takes place in the temperature range of approximately 500 to 900°C. It is a sluggish process and is not a problem during welding of the austenitic stainless steels, but can occur in elevated temperature service or if the welded component is stress relieved.
Formation of these phases is promoted by high chromium and molybdenum (ferrite forming elements) so that steels such as type 310 and type 316 are particularly sensitive and may show a substantial loss of ductility after stress relief. Delta ferrite also transforms more rapidly that austenite so those alloys containing large amounts of this phase will degrade faster than austenitic steel with only a small percentage of ferrite; hence the problems with duplex and super duplex stainless steels.
When it is necessary to stress relieve a fabrication then the loss of ductility must be accounted for. In those steels containing delta-ferrite, this phase should be held to a minimum, consistent with minimising the risk of hot cracking, by control of the ferrite forming elements by requiring typically 2% to 5% delta ferrite.
The phenomenon of embrittlement in austenitic stainless steel welds exposed to high temperature is accelerated by the presence of delta ferrite.
To avoid solidification cracking in austenitic stainless steel welds (see What is hot cracking/solidification cracking?), the composition of the filler material must be optimised to ensure that there is some delta ferrite present in the weld metal (typically >3%).
However, delta ferrite transforms into intermetallic phases, notably sigma phase, faster than austenite either during high-temperature service or during post-weld heat treatment (PWHT). Sigma phase is an intermetallic with an approximate chemical formula FeCr and, as with most intermetallics, it is very brittle and hence has a deleterious effect upon mechanical properties. It has been shown that, for a variety of iron-chromium-nickel alloys, Charpy toughness drops off exponentially with increasing sigma phase content.[1] The more delta ferrite a nominally austenitic stainless steel has, the more susceptible it will be to sigma phase formation. To avoid significant embrittlement it is typically desirable to limit the delta ferrite content in the original microstructure to below 10%.
Weld metal-containing delta ferrite will also be susceptible to ‘475°C embrittlement’. This phenomenon is observed when stainless steels are heated into the range 400 – 550°C (although the effect is most pronounced at 475°C) and a dramatic fall in toughness is observed after extended exposure. This is due to the formation of chromium-rich domains and precipitates within the iron-rich matrix by spinodal decomposition of ferrite at these temperatures. This effect becomes more pronounced as chromium content increases. However, 475°C embrittlement is not generally significant for materials with a ferrite number less than 14FN.

