
In Part 1, we know about Pitting corrosion. We will continue with remain one:
2) Crevice ( gap ) corrosion– Stainless steel requires a supply of oxygen to make sure that the passive layer can form on the surface. In very tight crevices, it is not always possible for the oxygen to gain access to the stainless steel surface thereby causing it to be vulnerable to attack.
Crevice Corrosion refers to the localized attack on a metal surface at, or immediately adjacent to, the gap or crevice between two joining surfaces. The gap or crevice can be formed between two metals or a metal and non-metallic material. Outside the gap or without the gap, both metals are resistant to corrosion.
Crevices can be:
- The space under a washer or bolt head.
- The gap between plates bolted together.
- The gap between components intermittently welded.
- The space under a sticky label.
- The space between a gasket and the metal in a flange (especially if the gasket is absorbent).
- Any other tight gap.
The mechanism: To be an effective corrosion site, the crevice must be wide enough to allow the corroding electrolyte in and then provide ‘stagnant’ conditions. Crevice corrosion can therefore be a concern where gaps are a few micrometers wide, but there are no absolute or critical dimensions for crevices, below which corrosion is certain.
As a corrosion mechanism, crevice attack is similar to pitting (i.e. free surface localized attack).
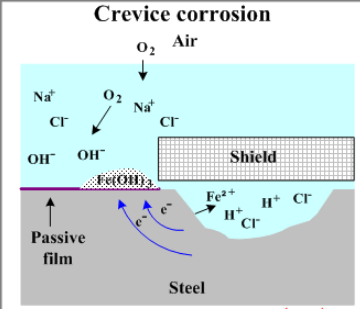
Why crevices can corrode:
Stainless needs free access to oxygen. Crevices are wide enough to permit entry of moisture, but narrow enough to prevent free circulation.
The result is that the oxygen in the moisture is used up. In addition, if chlorides are present they will concentrate in the stagnant conditions and, by a combination of reactions, the moisture can become acidic.
These are all conditions that can lead to the breakdown of the passive film on the stainless. Attack can then progress rapidly.
Factors influencing crevices / How to avoid crevise corrosion:
-
Alloy Resistance: Using a more corrosion resistant alloy gives less crevice attack. For example, in seawater at ambient temperature, crevices will form on 304 if there is a 0.9mm gap, on 316 if there is a 0.4mm gap and on 904L (similar corrosion resistance to 2205) if there is a 0.15mm gap.
The stability of the passive layer on stainless steels is promoted by chromium and supported by nickel. Although the chromium metal ion supports the anodic reactions in crevice corrosion more than iron or nickel, alloys with increasing chromium are better in resisting crevice corrosion. For given chromium level, austenitic stainless steels seem to resist attack better then the lower nickel ferritic types. Molybdenum and nitrogen have a very marked affect on increasing resistance, molybdenum assisting by arresting the rate of attack once depassivation has occurred and rapid attack is usually the next stage.
As a general rule stainless steels such as the 6% molybdenum austenitics and superduplex grades can be expected to give the best crevice corrosion attack resistance. As a guide some common stainless steels, rated in decreasing resistance to crevice corrosion, follows:
- 1.4547 and 1.4529 (6% Mo austenitic) plus 1.4501 1.4410 1.4507 (superduplex)
- 1.4462 (2205)
- 1.4539 (904L)
- 1.4401/1.4436 (316)
- 1.4301 (304)
- 1.4016 (430)
- Crevice Shape: The geometry of the crevice will influence its susceptibility to attack and the speed of progress. The narrower and deeper (relative to its width) a crevice is the worse attack will be.Metal to flexible plastic crevices tend to be narrower than rigid metal to metal gaps so metal to plastic joints provide more aggressive crevices.
- Environment: The more aggressive the liquid outside the crevice, the more likely it is that the crevice will be attacked.This is why crevice attack can be a problem in a salty swimming pool but not in a fresh water tank.In the atmosphere, crevices beside the sea give more problems than in rural environments. If the liquid outside the crevice is very oxidising, eg with bleach, hydrogen peroxide or ozone, then crevice attack will tend to be more severe.
- Temperature: Once the CCT is exceeded, then as with pitting corrosion, higher temperatures mean corrosion is more rapid. The rule of thumb is that a 10 degress C rise in temperature will double the corrosion rate. They will corrode faster because the temperature is consistently higher.


- Minimising the risk of crevice corrosion: Good design, fabrication and operating practices will anticipate and hence minimise crevice corrosion.
- + Design: Design to minimise the occurrence of crevices. If a crevice is a necessary part of a component’s design – can it be made wider?
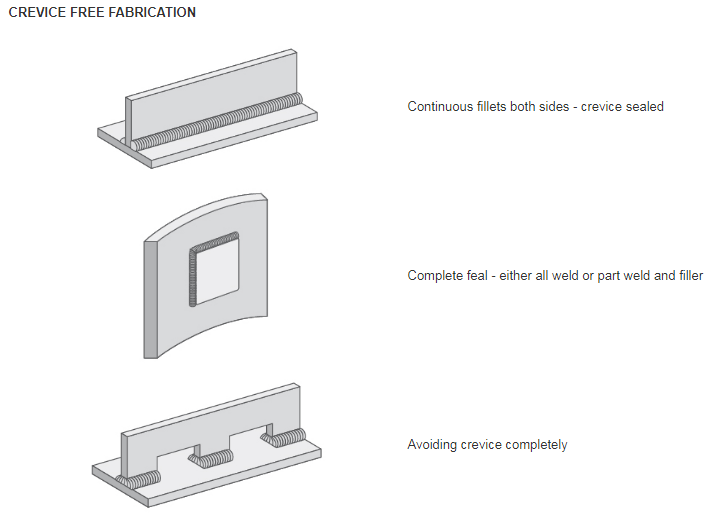
- Full penetration butt welds are best for joints. Seal lap joints and avoid gaps between pipes and fittings.Minimise use of bolted connections and other fasteners. Where crevices can’t be avoided use a steel grade resistant to crevice corrosion in the operating environment. It is also possible to seal the crevices to keep out corrosive liquids, but care must be taken that the seal is permanent. Be careful that the sealant ‘wets’ the surface. If it doesn’t it may form its own crevice. Sealants that dry and shrink can form their own crevices.Gaskets between flanges will probably form a slight crevice, but if the gasket does not absorb the liquid and is compressed between the surfaces (and not bulging around the flange), then the crevice is usually shallow enough so that crevice corrosion is not a problem.

- + Fabrication: Ensure full root penetration of welded joints with smooth weld bead. Avoid under cut and cracks in welding. Use of sticky labels or markers of various kinds (such as crayons) should be avoided, as should smears of grease or oil.

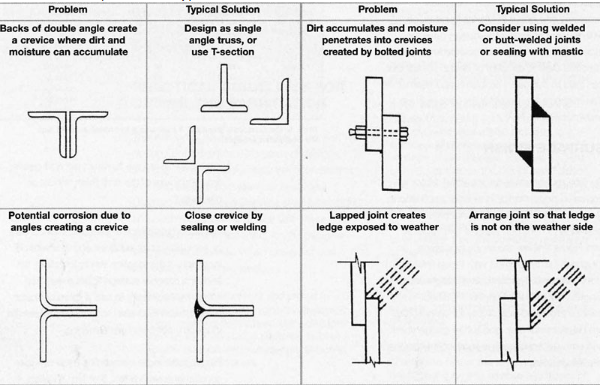
- + Operation: Sediment and scale can both result in crevices. If the problem can’t be designed out, routine maintenance will minimise risk. Crevice corrosion under bacteria film can occur. Maintaining circulation reduces the risk that debris will collect and form crevices in dead legs or low flow areas.
3) General corrosion- Normally, stainless steel does not corrode uniformly as do ordinary carbon and alloy steels. However, with some chemicals, notably acids, the passive layer may be attacked uniformly depending on concentration and temperature and the metal loss is distributed over the entire surface of the steel. Hydrochloric acid and sulphuric acid at some concentrations are particular aggressive towards stainless steel.
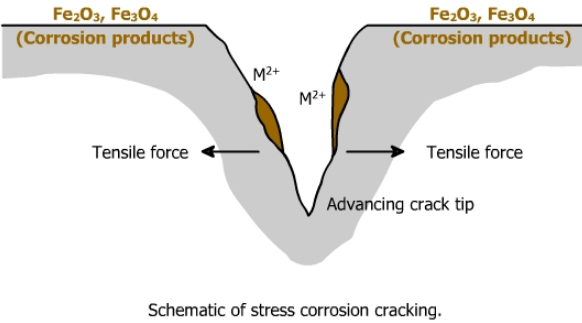
4) Stress corrosion cracking (SCC)– This is a relatively rare form of corrosion which requires a very specific combination of tensile stress, temperature and corrosive species, often the chloride ion, for it to occur. Typical applications where SCC can occur are hot water tanks and swimming pools. Another form known as sulphide stress corrosion cracking (SSCC) is associated with hydrogen sulphide in oil and gas exploration and production.

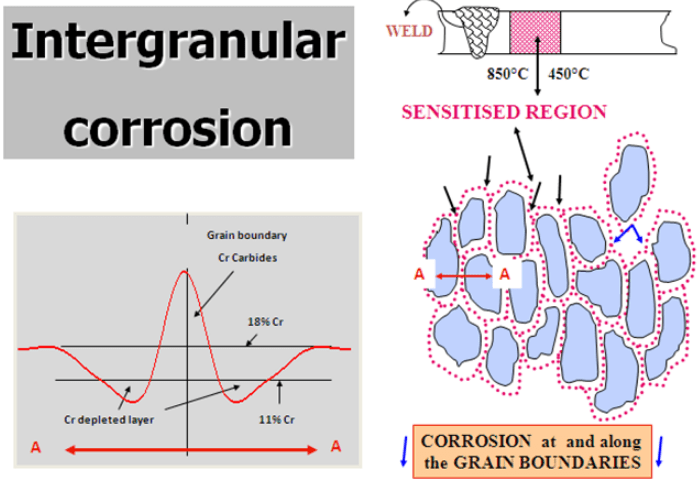
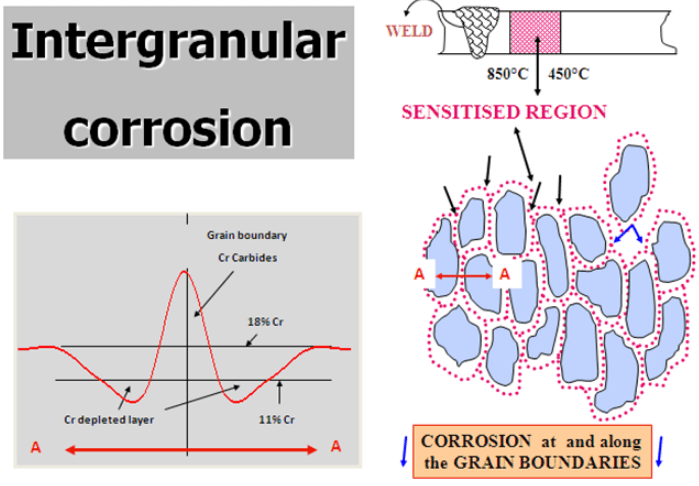
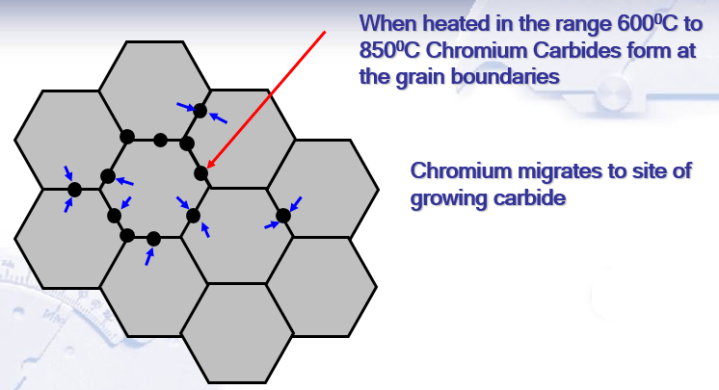
5) Intergranular corrosion ( weld decay ) – This is now quite a rare form of corrosion. If the Carbon level in the steel is too high, Chromium can combine with Carbon to form Chromium Carbide. This occurs at temperatures between about 450-850 deg C. This process is also called sensitisation and typically occurs during welding. The Chromium available to form the passive layer is effectively reduced and corrosion can occur. It is avoided by choosing a low carbon grade the so-called ‘L’ grades or by using a steel with Titanium or Niobium which preferentially combines with Carbon.


6) Galvanic corrosion- If two dissimilar metals are in contact with each other and with an electrolyte e.g. water or other solution, it is possible for a galvanic cell to be set up. This is rather like a battery and can accelerate corrosion of the less ‘noble’ metal. It can avoided by separating the metals with a non-metallic insulator such as rubber.

Conclusion: the passivation film on Stainless steel is very importance!!!


6 thoughts on “Why Stainless steel rust / corrosion? – Part 2 – Other corrosion.”