Selection and Treatment of Water and Conduct of the Test, to Maintain the integrity of the System is a priority concern in new-building or construction project. Specially on Stainless-steel which is very sensitive to corrosion.
1.0 Introduction
Welded fabrications are frequently tested hydrotested for pressure tightness. Unless properly conducted, this procedure can induce problems. It is important to avoid initiating corrosion of the fabrication by subjecting it to critical conditions outside the initial design constraints. Where the vessel is to handle materials which are later ingested, contamination must be avoided.

Damage may be caused by the induction of latent corrosion problems during and after hydrotesting. The damage may not become apparent until some weeks or months later, and may very difficult and costly to repair.
However, if simple precautions are taken, hydrotesting can be conducted without risk to the equipment. The precautions “mainly relate to the quality and the treatment of the water used, and how the equipment is treated after hydrotesting“. And hydrotesting even offers an opportunity to improve the quality of the fabrication during testing.
2.0 Corrosion of Stainless Steels
Corrosion failures of fabricated stainless steel systems soon after hydrotesting may result from one or more of three causes:
- Pitting & crevice corrosion (PCC)
- Stress corrosion cracking (SCC)
- Microbial induced corrosion (MIC)
API 570: Piping & Inspection Code, American Petroleum Institute.
Piping fabricated of or having components of 300 series stainless steel should be hydrotested with a solution made up of potable water (see note) or steam condensate. After testing is completed, the piping should be thoroughly drained (all high-point vents should be opened during draining), air blown, or otherwise dried. If potable water is not available or if immediate draining and drying is not possible, water having a very low chloride level, higher pH (>10), and an inhibitor addition may be considered to reduce the risk of pitting and microbiologically induced corrosion.
Note: Potable = ‘suitable for drinking’. Potable water in this context follows US practice, with 250 ppm maximum chloride, sanitized with chlorine or ozone.

Pitting and crevice corrosion can result from attack by the water used for hydrotesting. The equipment is designed for particular conditions, and the grade and surface finish are selected from knowledge of the proposed temperature, solute concentrations, pH etc. If the water conditions used for testing are more aggressive, corrosion may occur. Unfortunately, corrosion by these mechanisms is localized, and proceeds in isolation from the bulk solution conditions: once established, it can proceed in conditions which are otherwise not aggressive. Failure may occur by local penetration 3 – 12 months after initiation of the corrosion.
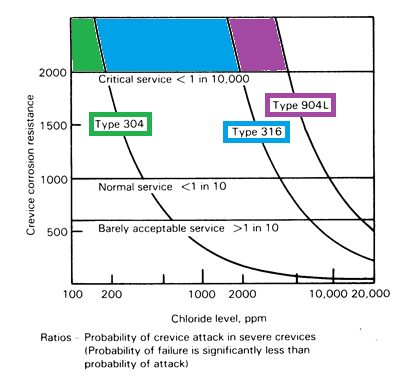
| The most aggressive ion in water is usually chloride, and accepted limits of this ion to avoid pitting & crevice corrosion are 200 ppm for grade 304L, and 2000ppm for 316(L), and 4,000 for duplex 2205. These values are not conservative. Note that some authorities limit the concentration of chloride to 100ppm. |
Time and temperature are also important in the development of pitting & crevice corrosion, and water with higher chloride content may be used safely if the contact time and temperature are limited.
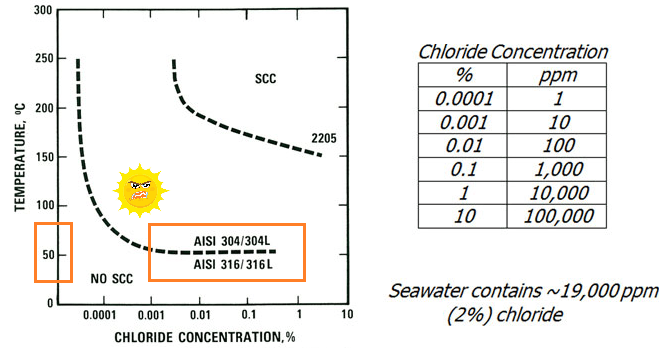
Stress corrosion cracking is a failure mechanism which can cause failure very rapidly. Stainless steels are not especially susceptible to stress corrosion failure in waters, but it may be encountered at high chloride levels and temperatures in austenitic grades such as 304 and 316. The duplex grades (e.g. 2205) are effectively immune from this form of failure in waters.
The austenitic grades are also effectively immune to stress corrosion cracking in waters at temperatures below about 50°C (figure 2). Since hydrotesting is usually performed at ambient temperature, the problem is unlikely to arise. However, equipment left in strong sunlight for a long period could get to this temperature, particularly in exposed pipe runs. Care should be taken to avoid excessive temperatures.
Microbiological Corrosion (MIC) is a mechanism which is increasingly being recognised as contributing to corrosion of metals. Penetration rates as high as 3.2 mm in a month have been reported for 304L, and in four months for 316L.
Micro-organisms, primarily bacteria, can alter the micro-environment to which the steel surface is exposed, by concentrating aggressive species, and by restricting the access of oxygen to the surface.
Sulphate reducing bacteria (anaerobic) metabolise sulphate ions to sulphide ions, while sulphate oxidising bacteria (anaerobic) can metabolise sulphur compounds to sulphuric acid. Both are highly aggressive to stainless steel.
The most common MIC problems with stainless steels are caused by iron utilizing bacteria, which concentrate chloride as iron and manganese chloride, to much higher concentrations than the level in the bulk of the water. This can easily far exceed the chloride levels mentioned above, and the resulting corrosion mechanism is often pitting and/or crevice corrosion: crevices, especially at gaskets, are one favored site for this form of corrosion. Another common site is at the weld. Numerous studies have shown that properly treated welds are not generally preferential sites for corrosion, but it is probable that surface imperfections such as oxide or slag inclusions, porosity.

MIC almost invariably occurs with raw (untreated) fresh water sources such as reservoirs, rivers, bores or wells, or fresh waters inadequately treated to prevent MIC.
Stagnant and low flow conditions promote attachment of bacteria to the metal surface. This is followed by colonization and formation of discrete deposits, starting as a biofilm, and frequently developing into a bulky deposit. The composition of the deposit is significantly different from and more corrosive than the bulk water. Localized attack manifests as:
- large subsurface cavities or tunnels, frequently at or adjacent to welds
- as broad, open pits in crevices such as gasketed flanged joints
- as broad conical pits with terraced sides
MIC can be prevented by draining and drying equipment after hydrotesting. The bacteria require time to grow, and no significant damage should be caused during the restricted time of hydrotesting.
This supposes that drying is complete and thorough, with no possibility of pools being left in the system. It is more conservative to also treat the water to disinfect it of bacteria before use.
Potable water supplies should be free of particulates, but other sources should be screened or settled and filtered. A filter passing particles up to 25 micrometers is probably adequate.
Sanitizations or disinfection of the water is essential to remove the risk of MIC. It can be carried out by a variety of methods, some of which also carry risks of making the water more aggressive for other forms of corrosion. Potable water from properly conditioned town supplies do not require further sanitization.
The most common sanitization method is chlorination, and the most common method of introducing chlorine is with a hypochlorite salt, which is available as a dry powder and as liquid solution, most conveniently from swimming pool supply shops. The addition required is usually small, and online additions are difficult to control. It is best, where possible, to disinfect the water as a batch before introducing it to the system under test, adding the disinfectant last and ensuring good mixing.
Free chlorine levels at entry to the system should be limited to 2ppm for grade 304, 5ppm for grade 316. The 2 ppm limit for residual free chlorine is also applicable to any copper alloys in the system.
Short term levels of free chlorine of up to 25 ppm can be tolerated by 316, provided there is thorough post cleaning treatment rinse, using water with the residual chlorine level maxima already mentioned.

After hydrotesting it is prudent to measure the chlorine residual of the last water to be drained from the system. If this is below 0.2ppm, or shows evidence of sludge, the system should be flushed with properly treated sanitized water without delay.
Other disinfectants which may be used include chlorine gas, chlorine dioxide, ozone and ultraviolet light. The former are subject to the same restrictions on free chlorine residuals, while ozone and ultraviolet light pose no risks for stainless steel. Indeed, ozone may be beneficial for corrosion resistance.

