To understand how marine corrosion protection works, it’s necessary to look in more detail in to the corrosion process. In this undesired chemical effect, the material can reach with different chemicals in its surroundings. The reaction can be divided into:
- Chemical reaction
- Electro-chemical reaction
These reaction take place exclusively at the surface of material. It’s possible the microscopic pits are formed by corrosion on the metal’s surface.The corrosion can also occur in existing crack.

CHEMICAL REACTION
In chemical reaction, there’s a charge transfer between the reactions. If the exchange of charge is local effect, then the resulting ” Chemical corrosion “.

An example of this reaction between Bare steel and air. A thin oxide layer formed at the surface rapidly. Then water come into contact to the oxide layer, the compound react (oxidation + reduction) to give the iron hydroxide (rust).
It is a very complex process which is completed in the following steps.
- Oxidation of iron-the iron gets oxidized into ferrous ions [Fe (II)] with the loss of two electrons.
- The ferrous ions again get oxidized into ferric ions [Fe(III)] in the presence of water and oxygen.
- These electrons from the above reactions are used to reduce oxygen.
- The ferric ions interact with oxygen and constitution ferric oxide [iron (III) oxide]. This ferric oxide gets hydrated with water.
The mechanism for the rusting process is similar to the electrochemical cell. The electrons formed during the oxidation of iron is conducted through the metal. Thus, the iron ions diffuse from the water layer to the metal surface where oxygen is present. This is an electrochemical cell where iron acts as the anode and oxygen gas as the cathode.
ELECTRO-CHEMICAL REACTION
Many compounds have the tendency to dissolve charged particle (ions) into water. Ion can move freely in water. Compounds that is always behave is this way are Acids, Alkaline, Soluble-salt, Metal and some Gases.
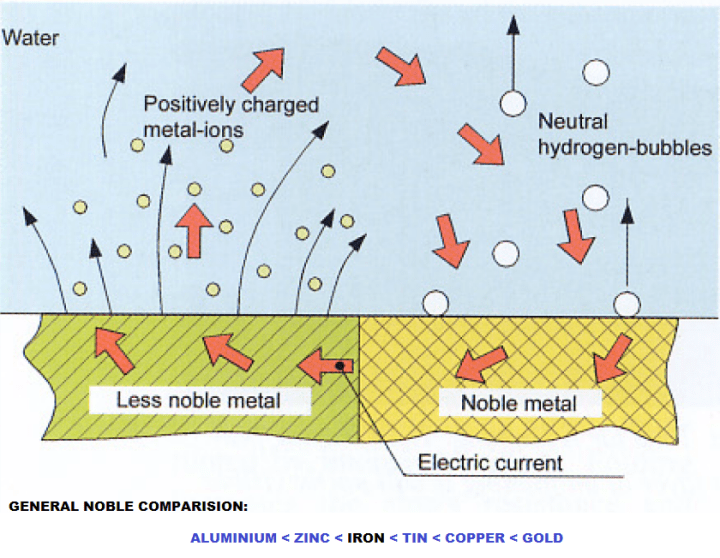
Electro-chemical corrosion can occur in the following places:
- Between Propeller & Surrounding Steel
- Between copper contained part ( heat exchanger ) and the steel part of piping system.
- Between aluminium/zinc parts and steel part of the ship.
Eliminating the corrosion current can prevent electro-chemical corrosion, it can be achieved in several way as below:
- Insulating the metal in the water by Painting.

- Reversing the current by using sacrificial anode (Zinc/Aluminium)

| TABLE I – GALVANIC SERIES OF METALS IN SEA WATER WITH REFERENCE TO SILVER/SILVER CHLORIDE REFERENCE CELL [Sea water flowing at 8 to 13 ft./sec. (except as noted), temperature range 50°F (10°C) to 80°F (26.7°C)] | |
| (ANODIC OR LEAST NOBLE) CORROSION-POTENTIAL | RANGE IN MILLIVOLTS |
| Magnesium and Magnesium Alloys | -1600 to –1630 |
| Zinc | -980 to –1030 |
| Aluminum Alloys | -760 to –1000 |
| Cadmium | -700 to –730 |
| Mild Steel | -600 to –710 |
| Wrought Iron | -600 to –710 |
| Cast Iron | -600 to –710 |
| 13% Chromium Stainless Steel, Type 410 (active in still water) | -460 to –580 |
| 18-8 Stainless Steel, Type 304 (active in still water) | -460 to –580 |
| Ni-Resist | -460 to –580 |
| 18-8, 3% Mo Stainless Steel, Type 316 (active in still water) | -430 to –540 |
| Inconel (78%Ni, 13.5%Cr, 6%Fe) (active in still water) | -350 to -460 |
| Aluminum Bronze (92% Cu, 8% Al) | -310 to -420 |
| Nibral (81.2% Cu, 4% Fe, 4.5% Ni, 9% Al, 1.3% Mg) | -310 to –420 |
| Naval Brass (60% Cu, 39% Zn) | -300 to –400 |
| Yellow Brass (65% Cu, 35% Zn) | -300 to –400 |
| Red Brass (85% Cu, 15% Zn) | -300 to –400 |
| Muntz Metal (60% Cu, 40% Zn) | -300 to –400 |
| Tin | -310 to –330 |
| Copper | -300 to –570 |
| 50-50 Lead- Tin Solder | -280 to –370 |
| Admiralty Brass (71% Cu, 28% Zn, 1% Sn) | -280 to –360 |
| Aluminum Brass (76% Cu, 22% Zn, 2% Al) | -280 to –360 |
| Manganese Bronze (58.8% Cu,39%Zn,1%Sn, 1%Fe, 0.3%Mn) | -270 to –340 |
| Silicone Bronze (96% Cu Max, 0.80% Fe, 1.50%Zn, 2.00% Si, 0.75% Mn, 1.60% Sn) | -260 to –290 |
| Bronze-Composition G (88% Cu, 2% Zn, 10% Sn | -240 to –310 |
| Bronze ASTM B62 (thru-hull)(85%Cu, 5%Pb, 5%Sn, 5%Zn) | -240 to –310 |
| Bronze Composition M (88% Cu, 3% Zn, 6.5% Sn, 1.5% Pb) | -240 to –310 |
| 13% Chromium Stainless Steel, Type 410 (passive) | -260 to –350 |
| Copper Nickel (90% Cu, 10% Ni) | -210 to –280 |
| Copper Nickel (75% Cu, 20% Ni, 5% Zn) | -190 to –250 |
| Lead | -190 to –250 |
| Copper Nickel (70% Cu, 30% Ni) | -180 to –230 |
| Inconell (78% Ni, 13.5% Cr, 6% Fe) (passive) | -140 to –170 |
| Nickel 200 | -100 to –200 |
| 18-8 Stainless Steel, Type 304 (passive) | -50 to –100 |
| Monel 400, K-500 (70% Ni, 30% Cu) | -40 to –140 |
| Stainless Steel Propeller Shaft (ASTM 630:#17 & ASTM 564: # 19) | -30 to +130 |
| 18-8 Stainless Steel, Type 316 (passive) 3% Mo | 0.0 to –100 |
| Titanium | -50 to +60 |
| Hastelloy C | -30 to +80 |
| Stainless Steel Shafting (Bar) (UNS 20910) | -250 to +60 |
| >Platimium | +190 to +250 |
| Graphite | +200 to +300 |

To be continued in MARINE COROSSION AND PROTECTION (Part 2)


2 thoughts on “MARINE COROSSION AND PROTECTION (Part 1)”